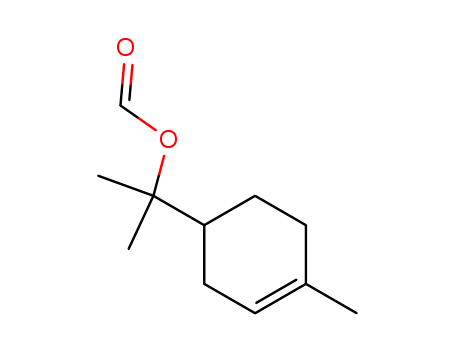
TERPINYL FORMATE
Base Information
- Chemical Name: TERPINYL FORMATE
- CAS No.: 2153-26-6
Export Top Purity TERPINYL FORMATE 2153-26-6 In Stock
- Molecular Formula: C11H18O2
- Molecular Weight: 182.263
- Vapor Pressure: 0.0469mmHg at 25°C
- Refractive Index: n20/D 1.471(lit.)
- Boiling Point: 236.6°Cat760mmHg
- Flash Point: 94.7°C
- PSA: 26.30000
- Density: 0.971g/cm3
- LogP: 3.32030
TERPINYL FORMATE(Cas 2153-26-6) Usage
|
Preparation |
By cold formylation of terpineol, using formic-acetic anhydride (Arctander, 1969). Uses: In public use before the 1920s. Use in fragrances in the USA amounts to approximately 10001b/yr. |
|
Synthesis |
May be produced from d-α-terpineol and formic acetic anhydride; the racemic α-terpineol is also obtained by an analogous synthesis. |
InChI:InChI=1/C11H18O2/c1-9-4-6-10(7-5-9)11(2,3)13-8-12/h4,8,10H,5-7H2,1-3H3/t10-/m1/s1
2153-26-6 Relevant articles
Discovery of a novel series of α-terpineol derivatives as promising anti-asthmatic agents: Their design, synthesis, and biological evaluation
Zhu, Wanping,Liu, Xia,Wang, Yuji,Tong, Yeling,Hu, Yongzhou
, p. 419 - 425 (2017/12/07)
A series of novel α-terpineol derivative...
Α- [...] enol derivative and its preparation method and application
-
Paragraph 0033; 0034, (2017/05/03)
The invention discloses alpha-terpineol ...
ANTIMICROBIAL COMPOSITION COMPRISING THYMOL AND A TERPINYL DERIVATIVE
-
Page/Page column 29, (2013/03/26)
The present invention relates to an anti...
P-Toluenesulfonyl chloride as a new and effective catalyst for acetylation and formylation of hydroxyl compounds under mild conditions
Khazaei, Ardeshir,Rostami, Amin,Mantashlo, Fatemeh
experimental part, p. 1430 - 1434 (2011/10/08)
The catalytic application of p-toluenesu...
2153-26-6 Process route
-
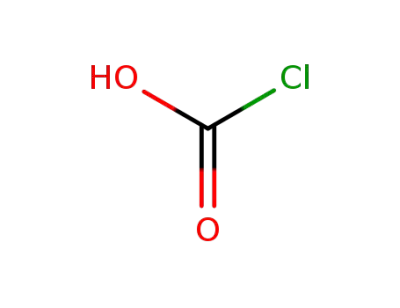
-
463-73-0
carboxy chlorine

-
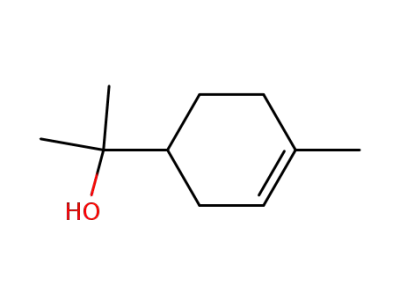
-
98-55-5,2438-12-2
terpineol

-
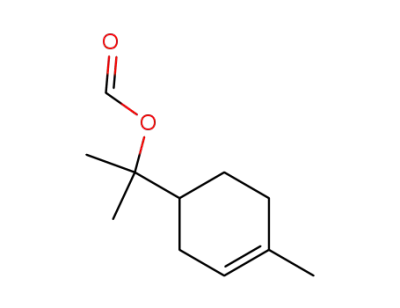
-
2153-26-6
2-(4-methylcyclohex-3-enyl)propan-2-yl formate
| Conditions | Yield |
|---|---|
|
With
sodium hydride;
In
tetrahydrofuran; mineral oil;
at 20 ℃;
Inert atmosphere;
|
-
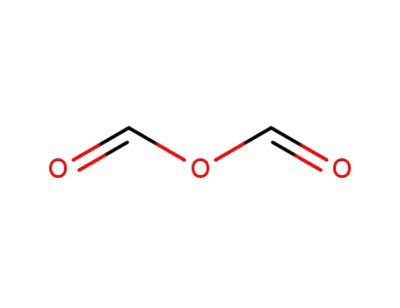
-
1558-67-4
formic anhydride

-

-
98-55-5,2438-12-2
terpineol

-

-
2153-26-6
2-(4-methylcyclohex-3-enyl)propan-2-yl formate
| Conditions | Yield |
|---|---|
|
With
p-toluenesulfonyl chloride;
at 20 ℃;
for 1.5h;
|
15.5%
|
|
With
p-toluenesulfonyl chloride;
at 20 ℃;
for 8h;
|
3.0032 g
|
2153-26-6 Upstream products
-
64-18-6
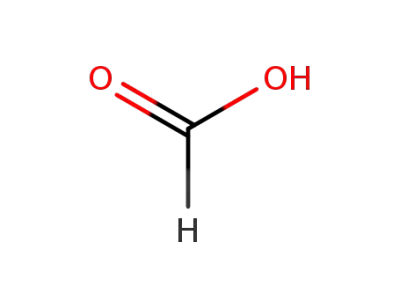
formic acid
-
39864-10-3
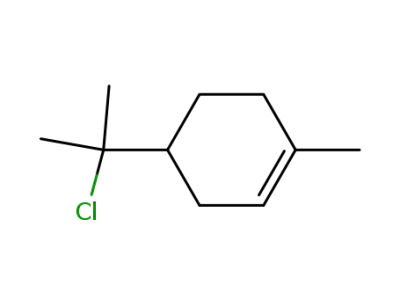
α-terpinyl chloride
-
2258-42-6
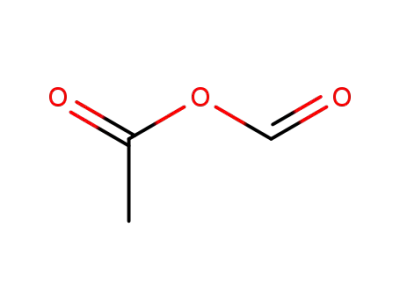
formyl acetic anhydride
-
138-86-3
limonene.
2153-26-6 Downstream products
-
38235-58-4
trans-sobrerol
Product recommendations
- 2,4-DIMETHYL-1,3-PENTADIENE
- (2,2-Diethoxyethyl)guanidinium sulphate
- 2-Methylhexadecyl acrylate
- Tetra-(methylethylketoxime)silane
- ETHYLENE GLYCOL HYDROXY STEARATE
- 1,3-dioxido-2,4-bis[[3-[(2,2,6,6-tetramethyl-4-piperidyl)oxy]propyl]amino]cyclobutenediylium
- N-Benzylglycine
- 3-[[4-(diethylamino)-o-tolyl]azo]-1,2-dimethyl-5-nitro-1H-indazolium acetate